Orally bioavailable BRD9 degrader CFT8634 and multi-channel risk

hERG Didn’t Kill This Program. Cardiac Biology Did
Orally bioavailable BRD9 degrader CFT8634 and multi-channel risk
Announcement



Full Text
Cardiotoxicity is not a niche liability; it is a core component of the safety profile for essentially every small-molecule program. Whether a compound is intended for oncology, inflammation, metabolic disease, or CNS, regulators and internal governance treat cardiac safety, especially proarrhythmic risk, as a non-negotiable gate. The practical consequence is simple: a drug candidate can be pharmacologically compelling, clinically active, and still become non-viable if its cardiac signal is not understood early and managed rigorously.
Despite this, critical assessment errors remain common. Too often, cardiac safety is reduced to one surrogate endpoint: hERG. If its inhibition by the compound is weak enough, the program moves forward. That simplification is operationally convenient and strategically expensive. It creates the illusion of control in preclinical development while leaving teams exposed to late-stage outcomes that look “unexpected” only because the evaluation framework was incomplete.
A concrete example is CFT8634, an orally bioavailable BRD9 degrader that advanced into a first-in-human Phase 1 study on the back of a strong preclinical package and apparent safety margins in GLP toxicology. In the clinic, however, the program encountered cardiac toxicities, including nonspecific T-wave abnormalities and QT prolongation, which ultimately constrained further development as monotherapy.
Notably, the team explicitly describes a disconnect between preclinical testing and human findings, underscoring the limits of standard preclinical cardiac models when the underlying risk drivers are not fully captured.
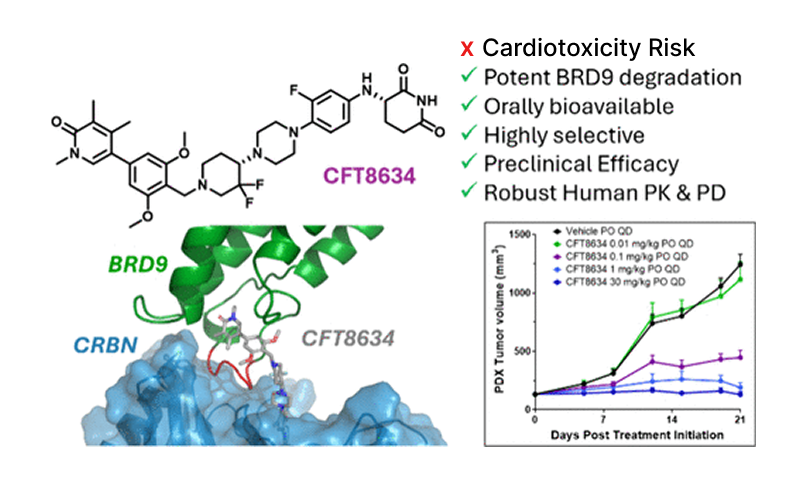
This is not bad luck. It is the predictable result of a widespread methodological mistake: treating hERG inhibition as the cardiac safety verdict. hERG matters, but it is not the whole story. Cardiac electrophysiology (including QT prolongation risk) emerges from multiple ion channels acting in tandem, with combined effects that cannot be reliably reduced to a single-channel proxy.
This multi-channel reality, and what to do about it, is where the technical discussion must begin.
How Multiple Ion Channels Jointly Drive Cardiotoxicity
QT prolongation is not a single-target effect. It emerges from how a compound reshapes the ventricular action potential through a coupled network of inward depolarizing currents and outward repolarizing currents. Treating “cardiac risk” as synonymous with one channel readout ignores the fact that the clinical phenotype is produced by the net effect across this system.
On the repolarization side, IKr (hERG) is important, but it is not acting alone. IKs and IK1 channels provide additional repolarizing capacity and stabilize terminal repolarization. On the opposing side, inward currents, particularly ICa,L (plateau) and late INa (INaL), sustain depolarization and can prolong repolarization when they are increased or left unopposed. Torsades de pointes (TdP) risk increases when outward repolarization is reduced without a compensatory reduction in inward currents, because that combination favors excessive QT prolongation and repolarization instability.
This is the operational meaning of repolarization reserve: the heart can buffer partial impairment of one current until the reserve is eroded by multi-channel drug effects, exposure, patient factors, or drug–drug interactions. As a result, two compounds with similar hERG inhibition can carry very different QT prolongation liability depending on their broader ion-channel fingerprint.
Building on this multi-channel reality, we use an approach designed to reflect it rather than simplify it away. CARDINAL (Cardiac Arrhythmia Risk Determination via Integrated Nonlinear Action-potential Logic) – part of the ADMETiQ model family – is Receptor.AI’s integrated cardiac safety platform that combines AI-based multi-channel IC₅₀ prediction with a mechanistic ventricular action-potential simulator. CARDINAL translates a compound’s full ion-channel inhibition profile into simulated electrophysiology, enabling direct assessment of the net impact on repolarization. In practice, this captures interaction-driven risk patterns that isolated assays cannot resolve and supports earlier, decision-grade prioritization ahead of clinical trials.
Like all preclinical cardiac safety frameworks, these predictions are grounded in assumptions about drug exposure at the tissue level. The concentrations used in CARDINAL represent effective cardiac tissue concentrations. In the in-vitro analogue, this corresponds to cells exposed to a solution at the specified concentration; in the human in-vivo context, it reflects the assumed concentration within myocardial tissue.
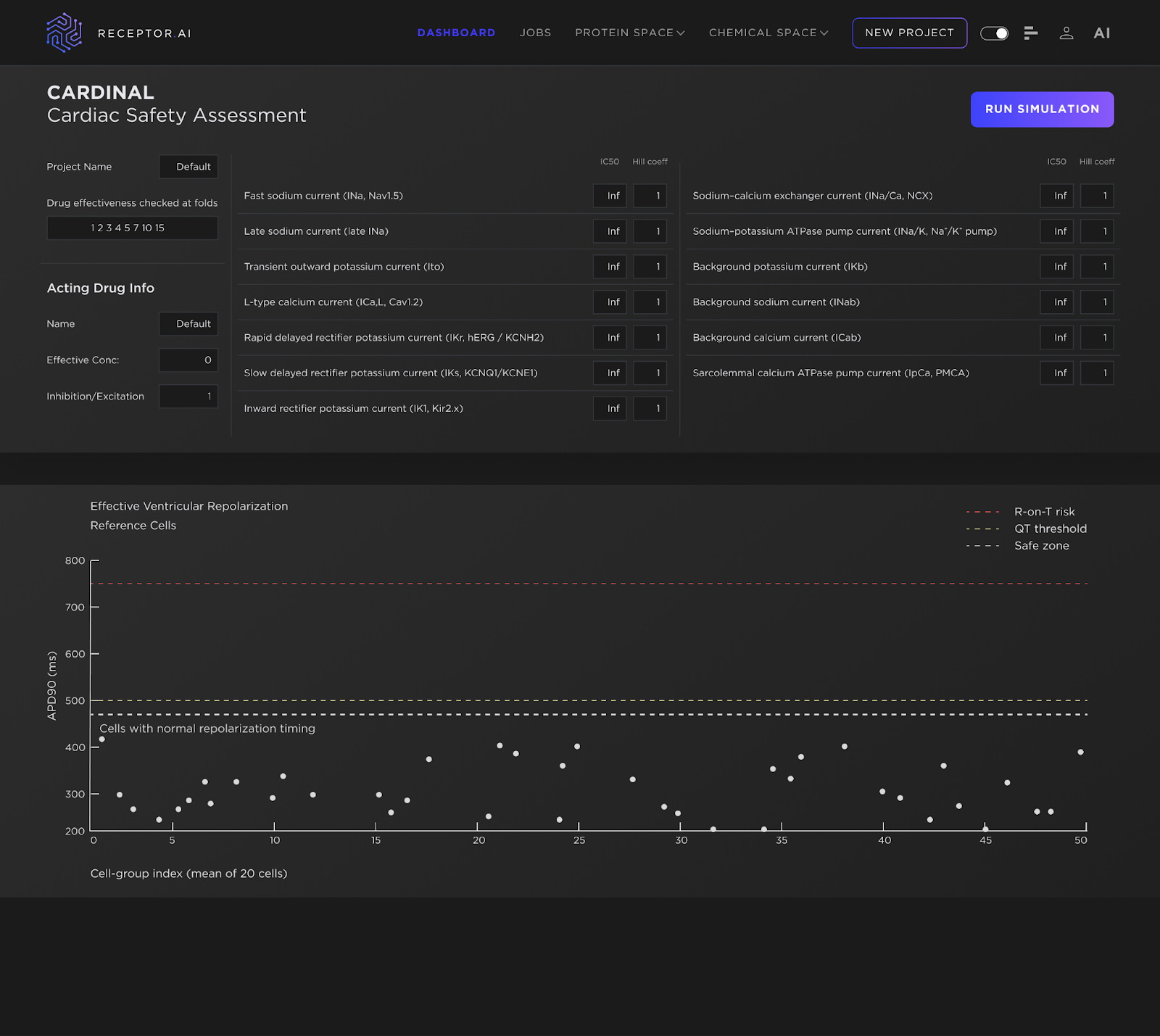
From hERG-Only to Multichannel Risk: What CARDINAL Surfaces
To make the value of CARDINAL concrete, we can model a counterfactual: how a program like CFT8634 might have been interrogated if it had been stress-tested with a systems-level cardiac readout before major preclinical and clinical investment.
Here we use APD-90 (action potential duration at 90% repolarization) as the primary readout. APD-90 is a direct measure of repolarization timing in ventricular cardiomyocytes and a practical proxy for QT-related liability: as APD-90 lengthens and approaches timing limits, repolarization reserve is being consumed and vulnerability to proarrhythmic conditions increases. As APD-90 lengthens further, repolarization encroaches on the available diastolic interval, creating timing conditions under which premature depolarizations, if they occur. are more likely to interact with vulnerable phases of repolarization. At the tissue level, this corresponds to an increased susceptibility to R-on-T–type events rather than direct observation of R-on-T at the cellular scale
Importantly, the multichannel perturbation scenarios explored here should be interpreted as mechanistically plausible stress conditions, not definitive statements about the exact inhibition profile of CFT8634. The intent is to probe regions of reduced repolarization reserve where modest, concurrent effects across several currents—each individually unremarkable and potentially below experimental detection thresholds—can combine into clinically relevant timing instability. This perspective is particularly relevant for heterobifunctional degraders, where indirect electrophysiological effects, proteomic remodeling, or downstream pathway modulation may influence ion-channel function in ways not captured by standard single-channel assays.
Under assumptions typical of standard in-vitro workflows, the compound appears well tolerated. The reference virtual population establishes the baseline APD-90 distribution (Figure 2), and a hERG-only perturbation at clinical exposure produces only modest changes that remain within nominal bounds (Figure 3). From a conventional, hERG-centric perspective, these results would be interpreted as acceptable.
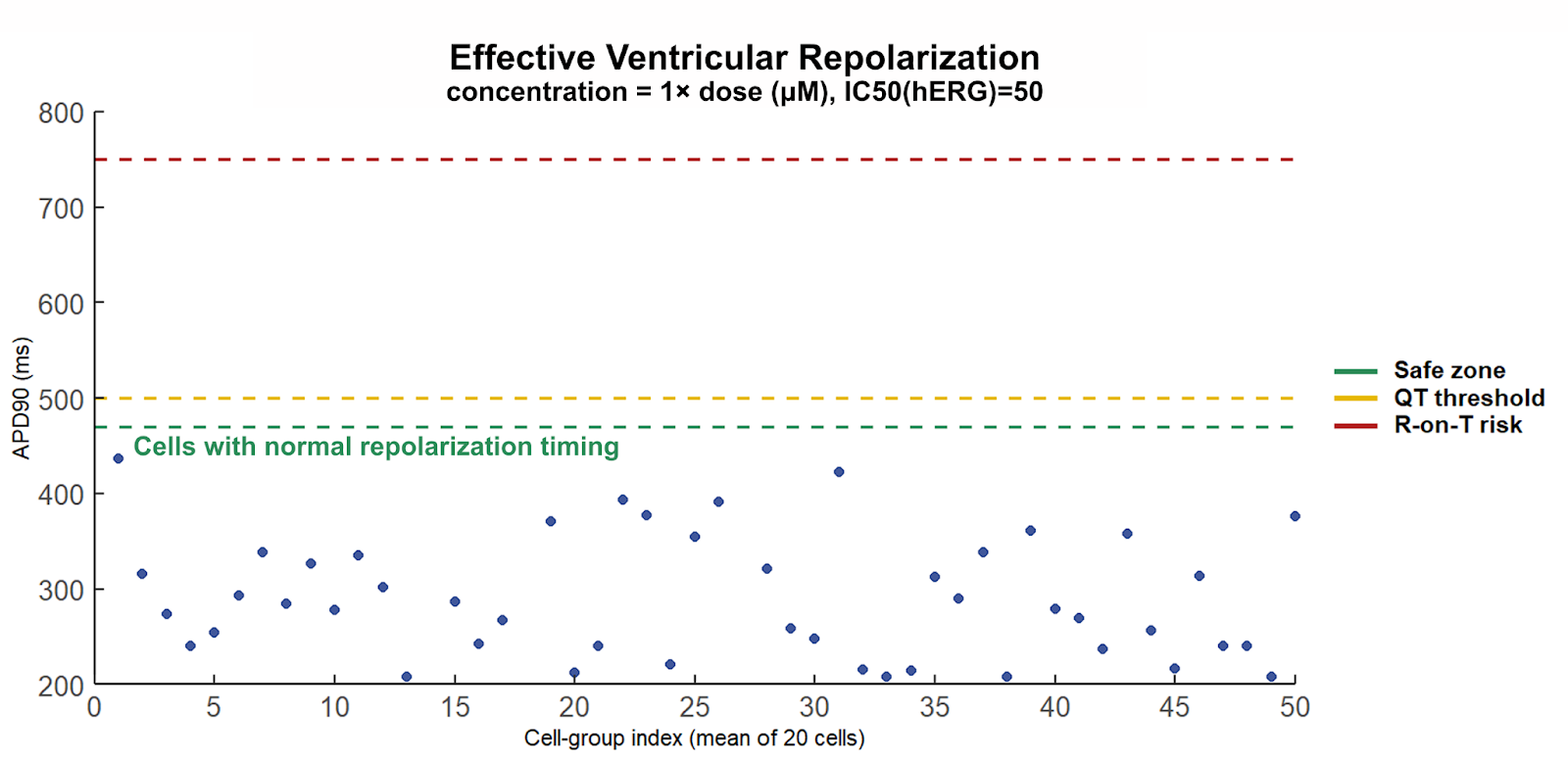
However, under supra-clinical exposure stress, APD-90 values progressively approach the upper boundary of the normal repolarization window (Figure 4). In practical terms, the system is already operating with critically reduced repolarization reserve, yet without an overt “fail” signal in hERG-only evaluation. This is the core preclinical blind spot: single-channel assessment can preserve the appearance of safety while the timing margin is being exhausted.
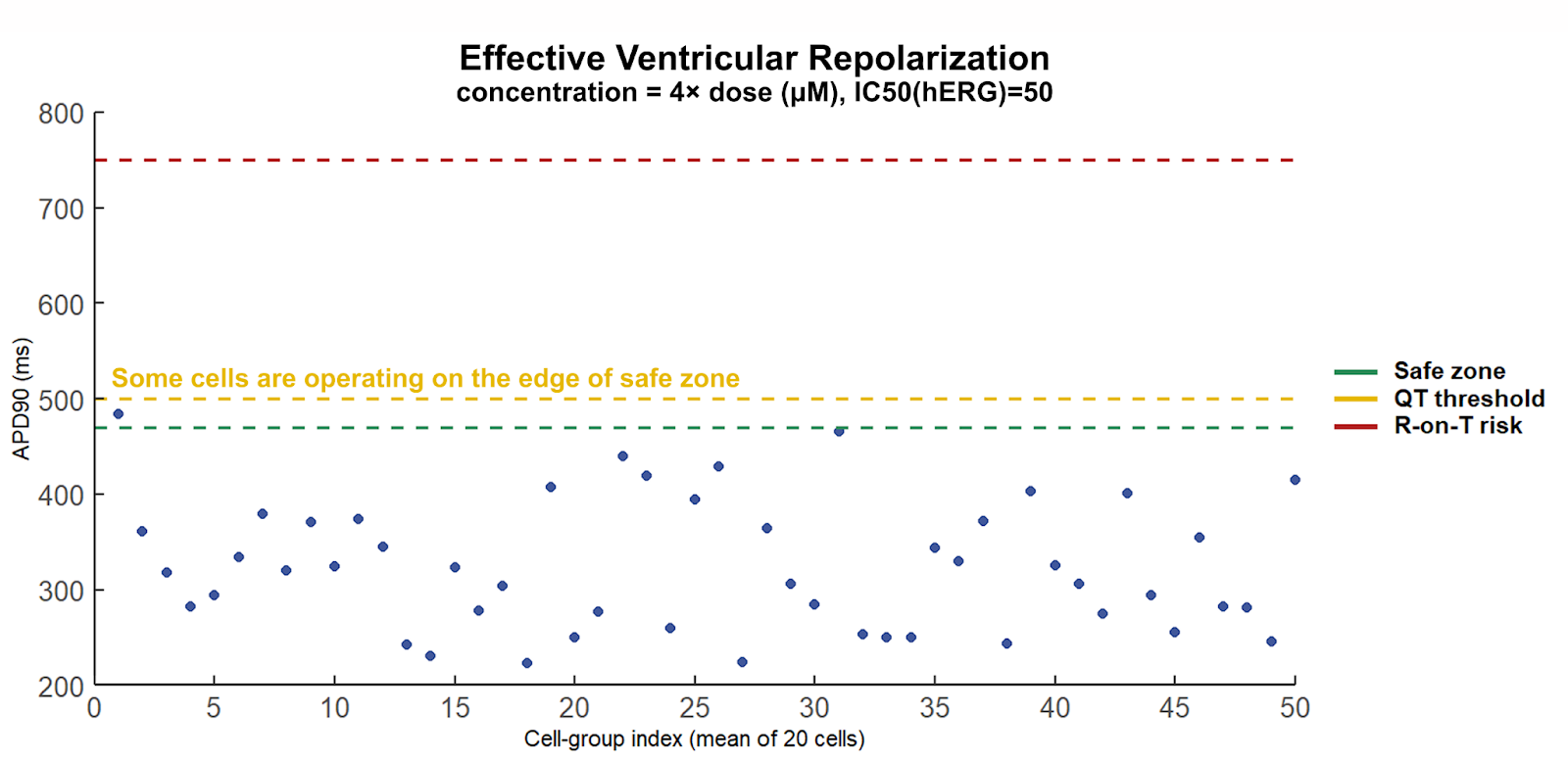
The picture changes decisively once additional ion channels are incorporated. Under the same supra-clinical exposure conditions, a distinct subpopulation emerges with markedly prolonged APD-90 values (Figure 5). Even when the population mean remains within nominal bounds, the appearance of heterogeneity is itself the signal. Cardiac safety is not governed by average behavior; the emergence of even a small subpopulation entering a prolonged repolarization regime indicates that the system has become timing-permissive for proarrhythmic events.
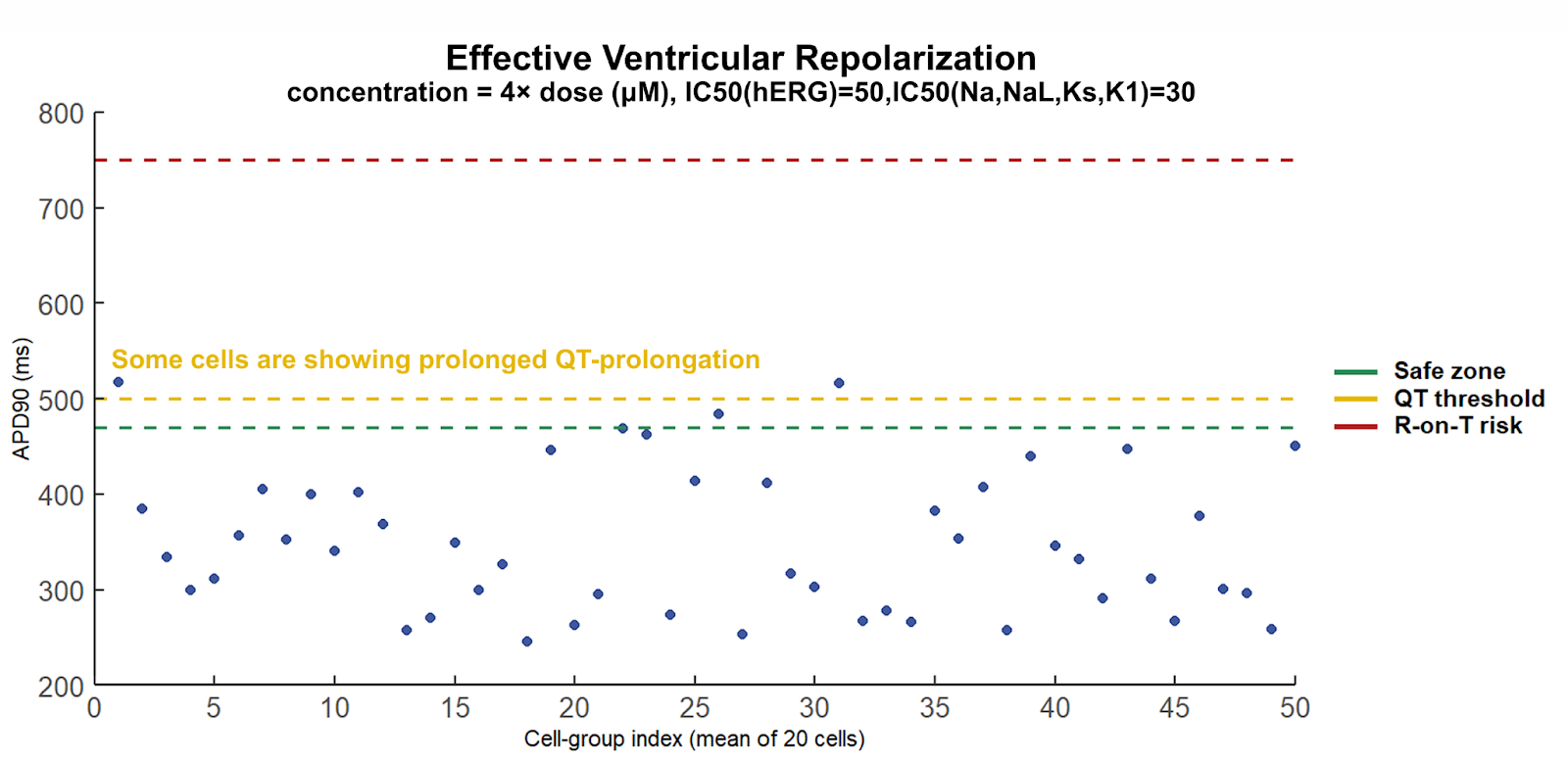
At higher exposure multiples, APD-90 lengthens further and the fraction of cells occupying high-risk timing regimes increases (Figure 6). This represents a timing-based susceptibility rather than a deterministic prediction of arrhythmia. Under these extreme conditions, APD-90 approaches or exceeds cycle lengths associated with normal resting heart rates, implying minimal or negative diastolic recovery time. This defines a repolarization overlap regime that is permissive for R-on-T–type triggers and TdP-associated mechanisms under routine perturbations such as pauses, ectopic beats, or heart-rate variability.
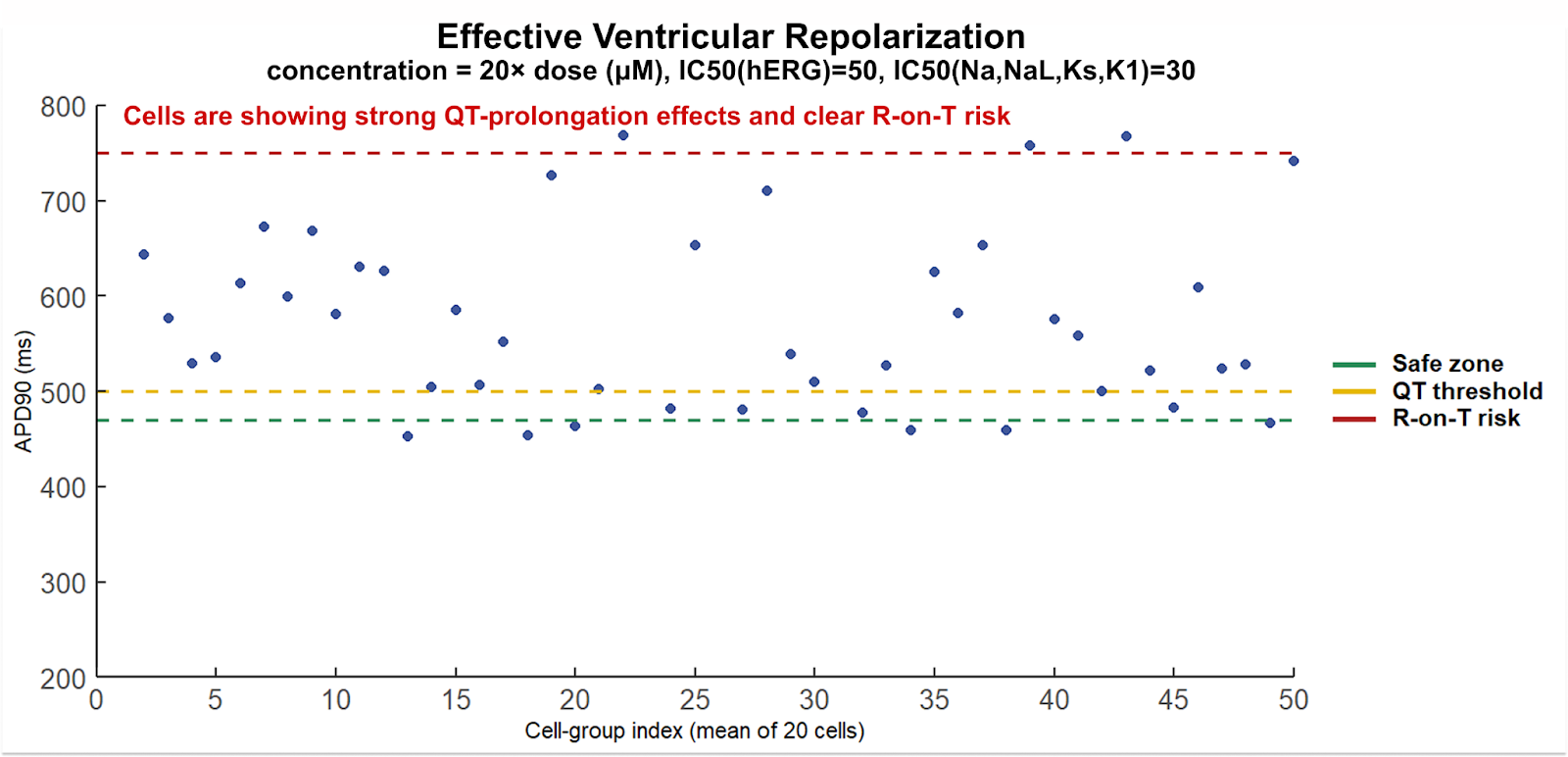
The takeaway is straightforward: a hERG-centric screening can look reassuring while the electrophysiological system is already operating near the edge, where modest multi-channel perturbations combine into a clinically meaningful proarrhythmic phenotype. CARDINAL can surface this liability when meaningful options still exist—compound redesign to rebalance the ion-channel profile, exposure and dose strategy adjustments, or proactive mitigation.
CARDINAL’s Application in Drug Discovery
CARDINAL is positioned as an early discovery screen, not a late-stage safety check. It can be applied when teams are still selecting which compounds to synthesize or purchase, and it remains useful as a parallel layer during hit-to-lead and lead optimization. If desired, CARDINAL can be run even before the first experimental ion-channel assays, using multi-channel IC₅₀ values predicted by Receptor.AI’s AI models as its input.
The practical outcome is a different decision process. Instead of advancing candidates on potency and a single hERG readout, teams can rank compounds by their simulated electrophysiological impact and prioritize those with a larger QT safety buffer under multi-channel interaction. This surfaces latent liabilities early, reduces downstream attrition risk, and gives medicinal chemistry a concrete optimization objective: shift the ion-channel profile toward electrophysiological stability while preserving on-target efficacy.
Conclusion
Cardiac safety failures are rarely “unexpected” in hindsight; they are usually the consequence of evaluating a system-level liability with a single-channel proxy. QT prolongation and torsades de pointes risk emerge from coordinated effects across multiple ion channels, where individually modest perturbations can combine into a clinically meaningful phenotype. The CFT8634 case illustrates the practical cost of relying on hERG-centric screening. A modern, decision-grade approach must integrate multi-channel pharmacology with mechanistic electrophysiology to surface risk early, when programs still have options to redesign molecules, optimize exposure, and mitigate liabilities. Receptor.AI’s CARDINAL is built to deliver that systems-level readout upstream, reducing late-stage surprises and improving the probability that clinically effective compounds remain clinically viable.

