
StratAI: Orchestrative Decision Engine for Drug Discovery
Announcement



Full Text
Drug discovery workflows are still commonly run as a rigid funnel, with a library screened using one method, compounds ranked, the top subset passed to the next method, and the same process repeated as chemical space is progressively narrowed. Once compounds have moved forward in the funnel, going back to rethink earlier decisions becomes difficult and expensive, often requiring substantial additional screening and validation.
This approach creates two main challenges. First, it can eliminate strong candidates because early-stage methods and scoring functions are imperfect, and compounds that look weak in a fast screen may prove highly promising under more accurate methods. Second, it does not scale to large chemical spaces, where screening often relies on clustering that further increases the risk of missing valuable compounds.
As a result, the system does not truly learn. It recalibrates scores within a narrowed chemical space, rather than using new evidence to question earlier assumptions, recover overlooked candidates, or decide which method should be applied next. This is why, at Receptor.AI, we built StratAI as a unified decision platform for small molecules, peptides, and induced-proximity modalities.
Decision engine for adaptive drug discovery workflows
StratAI is designed to navigate extremely large chemical spaces and move programs toward the best compounds more efficiently than a linear screening workflow. In a conventional funnel, compounds are advanced through the same fixed sequence of steps regardless of what has already been learned, which makes exploration expensive, slow, and often inefficient. In contrast, StratAI continuously decides which compounds to evaluate next, with which methods, and for what purpose, maximizing information gain while keeping resource use aligned with program goals.
This decision engine combines two core capabilities: uncertainty mapping and next-experiment selection. Candidates are evaluated against defined and customizable objectives using computational methods that estimate both predicted scores and the confidence associated with each prediction. Uncertainty mapping identifies where the model is reliable, where evidence is still weak, and where additional data would most affect the decision. Next-experiment selection then uses that information to choose the assay, simulation, or measurement most likely to reduce uncertainty and improve the next progression decision. Light methods are applied across candidates with low uncertainty for fast evaluation, while heavier methods are reserved for cases where uncertainty is high enough to obscure the decision-making process. Instead of following a fixed sequence of assays, StratAI adaptively chooses the experiments with the highest expected decision value.
The role of the decision engine, therefore, is to orchestrate computational and experimental methods, assign compounds to the appropriate level of evaluation, and adapt the workflow as new evidence emerges. Additionally, this functionality is not limited to a single modality, but can cover several, such as small molecules and peptides. Figure 1, showcases the implementation of StratAI for peptide optimization.
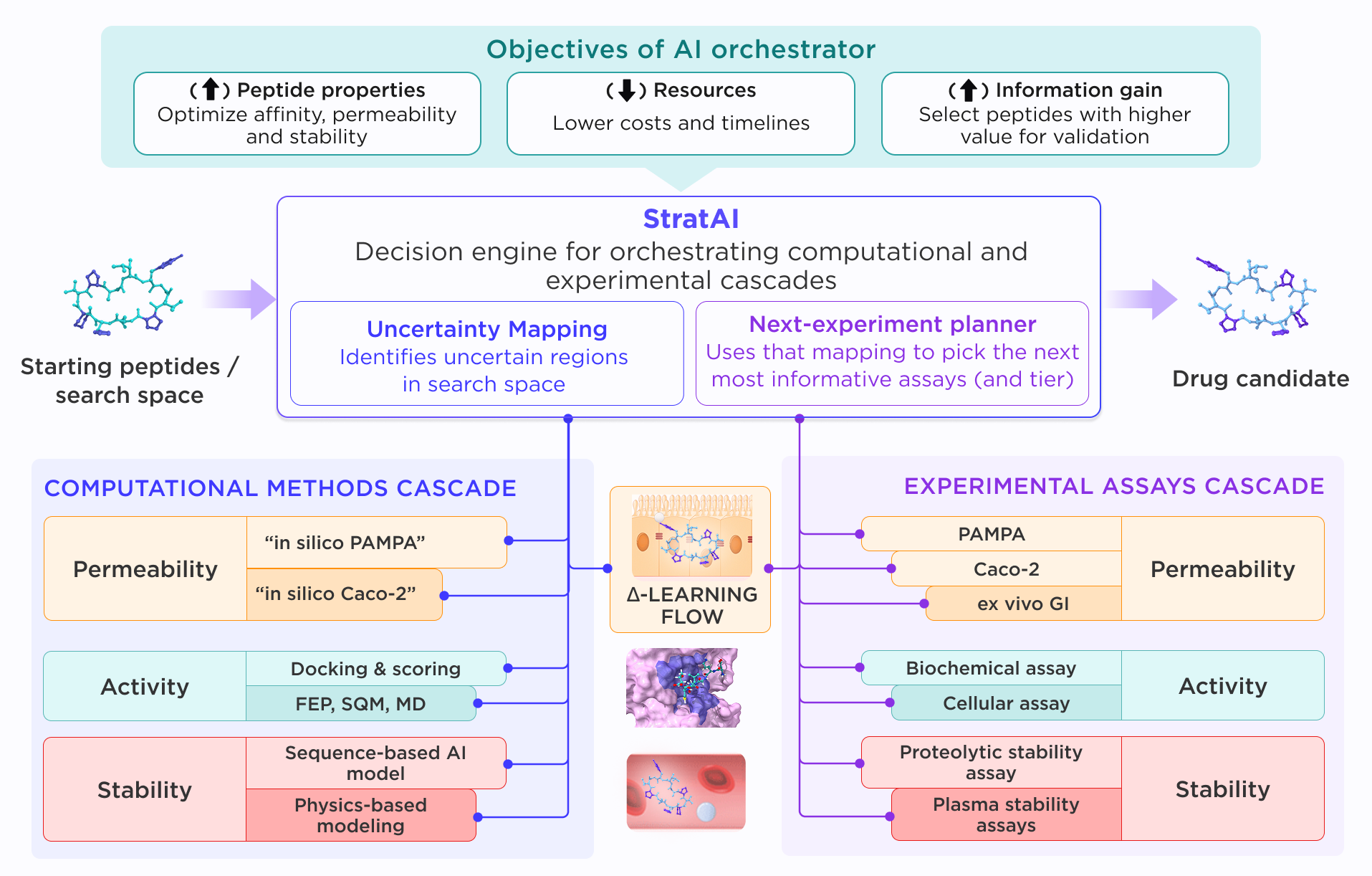
This flexibility is critical in drug discovery, where programs are defined by multiple objectives and often require unique combinations of computational and experimental tools. As those objectives evolve, the methods used to evaluate and prioritize compounds must evolve with them. StratAI is designed specifically for this setting, to help drug discovery programs evolve across multiple goals, not just a single endpoint.
Another key advantage of this approach is that StratAI does not attempt to screen chemical space exhaustively. Instead, it learns SAR/QSAR signals from accumulating computational and experimental data and uses that signal to propose the next compounds or modifications to test. Candidate selection is driven by expected improvement, uncertainty, and diversity, allowing the system to balance progress toward the objective with exploration of informative new regions of chemical space. The result is an adaptive workflow that navigates chemical space efficiently instead of applying brute-force screening across an unmanageably large space.
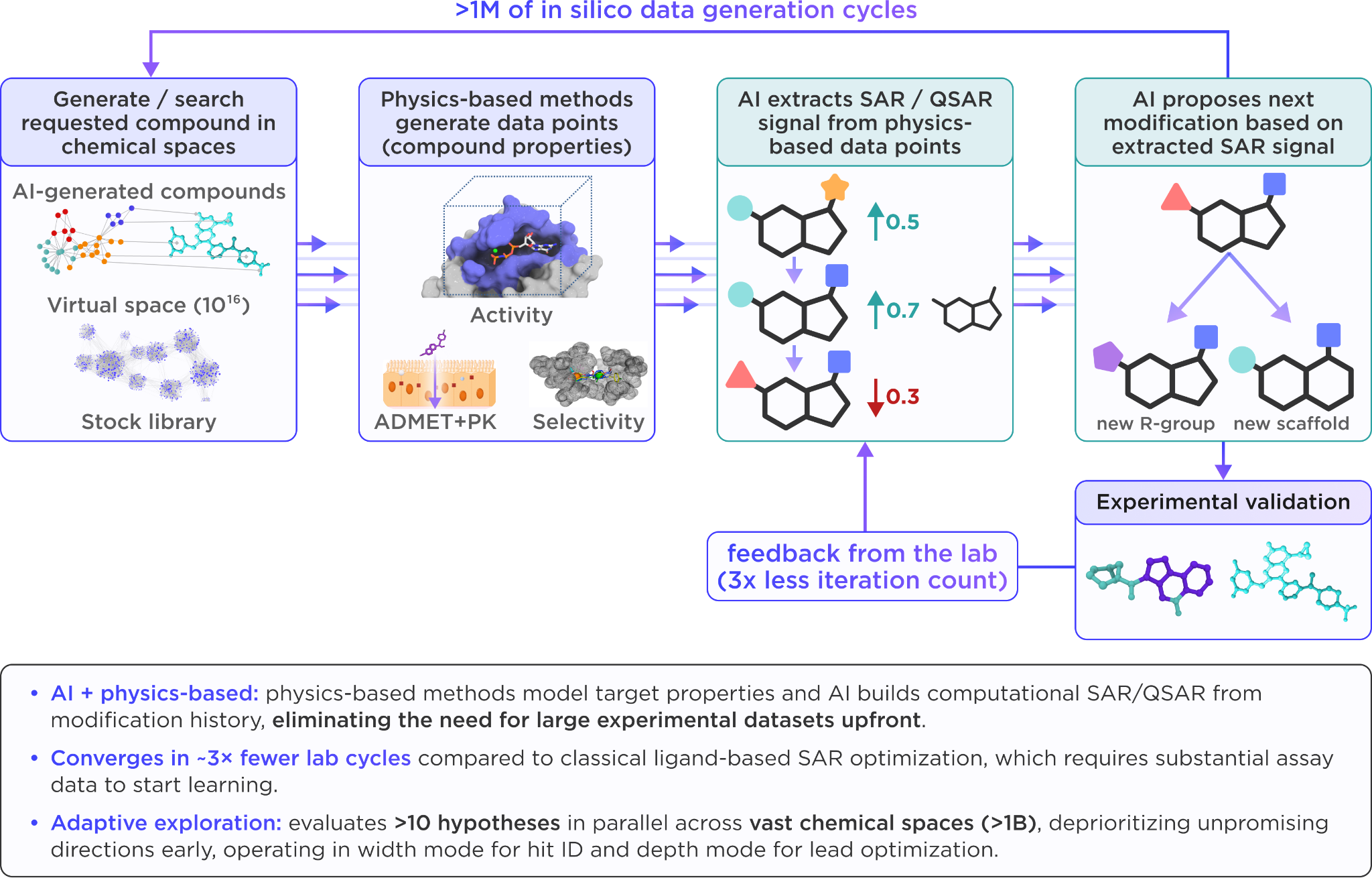
Moreover, StratAI is designed as a closed-loop system, where the results of each run are preserved in a structured form so that the reasoning of previous recommendations remain available for future runs. When a new program shares relevant characteristics with an earlier one, this accumulated knowledge can be transferred forward rather than rebuilt from scratch.
What lies in the intersection between StratAI and R&D?
QuorumMap operates at the intersection of R&D strategy and day-to-day execution. At the strategic level, drug discovery programs define shifting objectives across potency, selectivity, developability, and modality-specific constraints. At the operational level, teams must decide which compounds to prioritize, what evidence is sufficient for progression, and where limited experimental and computational capacity should be spent next. StratAI connects these two layers by turning program goals into an explicit decision framework for progression.
In this role, StratAI serves less as a scoring tool and more as operational decision infrastructure for R&D. It helps translate program priorities into explicit progression criteria, align computational and experimental teams around a shared decision logic, and preserve the rationale behind why compounds were advanced, and deprioritized. QuorumMap improves R&D efficiency by reducing unnecessary testing, shortening iteration cycles, and making progression decisions more reproducible across teams and programs.
Furthermore, StratAI supports multiple therapeutic modalities, including small molecules, peptides, and induced proximity programs, and applies a shared decision framework. Within this framework, StratAI can optimize core objectives such as affinity or selectivity alongside modality-specific parameters that are critical in each setting, such as permeability in peptides.
Summary
StratAI combines adaptive screening, selective experimental validation, and iterative learning in a unified decision framework for drug discovery. By using each cycle of evidence to guide the next, it enables more efficient navigation of chemical space, reduces unnecessary validation, and supports better progression decisions across modalities. In the future, platforms built on this logic could help reshape drug discovery by making optimization faster, more scalable, and better suited to the growing complexity of modern therapeutic programs.

